Surgeons in over 70 countries utilize MicroPort Orthopedics Hip & Knee products. Our national sales team is ready to answer any question you have.
MicroPort Orthopedics provides an Implant Card with each device. The intention of the Implant Card is to enable the patient receiving the implant to:
b. Identify themselves as requiring special care in relevant situations (i.e., security checks, emergency situations, etc.)
In meeting this intent, MicroPort Orthopedics requests the healthcare provider complete the following instructions in order to provide the patient with a fully compliant Implant Card.
United States: 866.872.0211
ii. Email: complaints@ortho.microport.com
• Reporter’s Address
• Reporter’s Phone Number
• Product Number
• Lot Number
• Detailed description of the incident
 | Patient’s name |
 | Date of surgery |
 | Name and address of doctor or healthcare center |


| Symbols Glossary | ||
| Symbol Title | [Additional Explanation] Reference Number/ Standard | Explanatory Text from Standard |
 | Medical device | Indicates the item is a medical device |
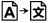 | Translation | Indicates that the original medical device information has undergone a translation which supplements or replaces the original information |
 | Batch code | Indicates the manufacturer’s batch code so that the batch or lot can be identified. |
 | Catalogue number | Indicates the manufacturer’s catalogue number so that the medical device can be identified. |
 | Manufacturer | Indicates the medical device manufacturer. |
 | MR Conditional | Indicates an item that has been demonstrated to pose no known hazards in a specified MR environment with specified conditions for use. If applicable, the person implanted with an MR Conditional medical device can safely undergo an MR exam only under very specific conditions. Scanning under different conditions may result in injury, severe injury and/or death or device malfunction. Full MRI safety information is available in the MRI Safety Information section of the product package insert, which can be obtained at www.ortho.microport.com/ifus or by calling +1.866.872.0211 or +44 1244.572120. |
 | Unique Device Identifier | Indicates a barcode as containing Unique Device Identifier information |
 | Patient identification | Indicates the identification data of the patient. |
 | Patient information website | Indicates a website where a patient may obtain additional information on the medical product. |
 | Health care center or doctor | Indicates the address of the health care center or doctor where medical information about the patient may be found. |
 | Date | Indicates the date that information was entered or a medical procedure took place. |
